Often times, I have come across websites, such as "AnswersInGenesis" (Judging from the title, it is obvious that this website isn't really about Science), which shamelessly debunk the basic tenets and empirical methods of Science.
Being the intellectual equivalent of hucksters and mobsters, Creationists of these sort are apt to criticize the Scientific Method in a bid to construe evidence to fit into their biblical bullshit. They thraw through scientific literature, pick the parts that supposedly criticize a scientific method, and put up claims that mainstream Science is bogus. These deranged fundamentalists are not interested in Science; they don't carry out any field work or research. All they ever do is throw around their banter and how that someone, somewhere, will lap up and believe in their God-induced beliefs, which coincidentally, have not scientific merit whatsoever.
So, without further ado, I would like to proceed to one of the more controversial topics amongst the Creationist community (not the Scientific Community): Carbon Dating.
Long regarded with as much vitriol as Darwin's revolutionary idea of Evolution, carbon dating debunks the "Young Earth" Creationist Model: Because of its ability to date fossils upwards to 60,000 years, radiocarbon dating has long been regarded as Darwin's (Read Satan's) advocate.
History Of Radio-Carbon Dating
Discovered by Willard Frank Libby and his colleagues in 1949, during his tenure as a professor at the University of Chicago, Libby invented what would eventually change the face of archaeology by giving a time frame to fossilized specimens.
Libby estimated that the steady state radioactivity concentration of exchangeable carbon-14 would be about 14 disintegrations per minute (dpm) per gram.
In recognition of his work, he was awarded the Nobel Prize in chemistry in 1960.
The Mechanics of C-14
Being the intellectual equivalent of hucksters and mobsters, Creationists of these sort are apt to criticize the Scientific Method in a bid to construe evidence to fit into their biblical bullshit. They thraw through scientific literature, pick the parts that supposedly criticize a scientific method, and put up claims that mainstream Science is bogus. These deranged fundamentalists are not interested in Science; they don't carry out any field work or research. All they ever do is throw around their banter and how that someone, somewhere, will lap up and believe in their God-induced beliefs, which coincidentally, have not scientific merit whatsoever.
So, without further ado, I would like to proceed to one of the more controversial topics amongst the Creationist community (not the Scientific Community): Carbon Dating.
Long regarded with as much vitriol as Darwin's revolutionary idea of Evolution, carbon dating debunks the "Young Earth" Creationist Model: Because of its ability to date fossils upwards to 60,000 years, radiocarbon dating has long been regarded as Darwin's (Read Satan's) advocate.
History Of Radio-Carbon Dating
Discovered by Willard Frank Libby and his colleagues in 1949, during his tenure as a professor at the University of Chicago, Libby invented what would eventually change the face of archaeology by giving a time frame to fossilized specimens.
Libby estimated that the steady state radioactivity concentration of exchangeable carbon-14 would be about 14 disintegrations per minute (dpm) per gram.
In recognition of his work, he was awarded the Nobel Prize in chemistry in 1960.
The Mechanics of C-14
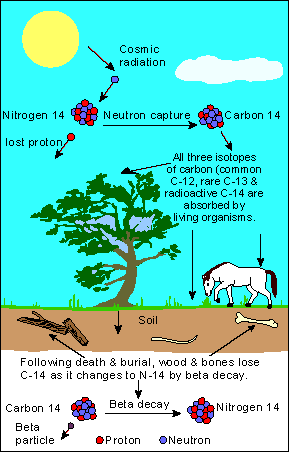
Before I begin to illustrate the basics of carbon dating (otherwise referred to as radiocarbon dating), it is prudent to explain how Carbon-14, the carbon compound that serves as a testing material for dating carbonaceous, fossilized matter.
Everyday, cosmic rays enter through Earth's atmosphere, bringing about certain chemical reactions which are essential for the sustainence of life on this planet.
When a cosmic ray collides with an atom in the atmosphere, a secondary cosmic ray is formed in the form of an energetic neutron.
When this neutron collides with a nitrogen atom (N-14), a chemical reaction takes place: The Nitrogen 14 (seven protons, seven neutrons) converts into a carbon-14 aom (six protons, eight neutrons) and a hydrogen atom.
Why C-14?
Because C-14 is constantly being absorbed by plants for the purpose of photosynthesis, the natural cycle of C-14 (Radioactive Carbon), along with C-12 (non-radioactive carbon), is constantly absorbed to other animals via the food chain.
Every living plant and animal has roughly the same ratio of C-14 to C-12, which is about 1: 1 trillion. As such, the absorption rate is fairly constant, that is, until death starts to kick in.
When an animal dies, the animal stops absorbing carbon, for the obvious reason that it no longer can consume food. At this point, the C-14 atoms begin to decay, while the C-12 atoms remain constant.
Because of C-14's ability to decay in contrast to C-12's constant, a prediction can be made towards the age of a fossil.
Characteristics of C-14
One of the most important properties of C-14 is its half-life: A half-life refers to the time whereby the number of a certain atom disintegrates or decays by half its original number.
Radiocarbon C-14 has a half-life of about 5,700 years. In short, it takes 5,700 years to reduce C-14 by 50%, another 5700 years to reduce to 25%, and so on, at an exponential rate.
The formula for C-14 dating is:
t = [ ln (Nf/No) / (-0.693) ] x t1/2
-ln is the natural logarithm;
-Nf/No is the percent of carbon-14 in the sample compared to the amount in living tissue;
-t1/2 is the half-life of carbon-14 (5,700 years).
Problems Faced Using C-14 Dating Method, & Ways To Overcome Them
The truth is, C-14 decomposition is highly consistent, with a margin error of plus or minus 40 years. That, in scientific parlance, is a very good tolerance.
The real problems of C-14 and its so-called erroneous readings, however, have more to do with the natural elements than the consistency of its decay.
1. Exponential Decay
One of the more severe problems has to do with exponential decay: Because of its ability to decompose, most C-14 work can only be carried out on objects with a maximum age of 60,000 years, as counting decayed C-14 for older objects become more arduous due to the extreme lack of C-14 atoms.
In short, the older the object, the wider the discrepancy. Any fossil more than 60,000 yrs old would have too little C-14 to carry out any meaningful C-14 dating.
In order to circumvent this problem, Accelerator-based mass-spectrometric (AMS) techniques was introduced: All the 14C atoms can be counted directly, rather than only those decaying during the counting interval allotted for each analysis. The AMS technique allows one to date samples containing only a few milligrams of carbon.
2. Varying C-14 Compositions
Variations of C-14 compositions in the atmosphere (Up to 1%, according to Hessel de Vries, a Dutch physicist at the University of Groningen).
3. Natural Causes
Certain natural phenomenon, such as changing climates, can affect C-14 flow in the atmosphere, hence affecting absorption rates by plants and animals.

In order to resolve this discrepancy within a certain tolerance, calibration charts are often used to mediate the readings. Various calibrations may include distortions caused by magnetic disturbances or abnormally high C-14 concentrations found in the vicinity of the fossil's environment.
4. Man Made Causes
C-14 concentration can also be affected by human activities:
i. It was almost doubled for a short period due to atomic bomb tests in the 1950s and 1960s.
ii. C-14 concentration was drastiscally reduced by the release of large amounts of CO2 from ancient organic sources where C-14 is not present (i.e Fossil fuels). This was known as the Suess Effect.
For this reason, raw, uncalibrated readings before 1950 are classified under BP (Before Present), which indicates the number of years of an object in relation to the 1950 date. This was the date given in part because of the absence of large-scale nuclear testing, particular in the Northern Hemisphere, which increased atmospheric C-14 dramatically.
Conclusion
Radio-carbon dating has been shown to be pretty consistent, once external factors, such as environmental carbon content are pretty much factored in and calibrated into the readings.
While Creationists keep harping about the inconsistency of Carbon Dating, they tend to "negate" that Science has kept up with the times: Much has been known about C-14 levels and its interactions with the natural processes to provide a more accurate intepretation of C-14 data, and advanced techniques such as the AMS has dramatically assisted in providing more accurate readings.
If Creationists and other religious fundamentalists want to play in the real world of science, they'd better do some real research before babbling their bullshit and pandering them around as Science.

Sure they can build their fancy Creationist Museums with their saddled Triceratops and some really impressive computer animatronics (no thanks to Hollywood animationists who sold their national conscience to rich fraudsters), but when it comes to real Science, I will suggest that they let the real scientists do the real work instead.




61 comments:
that was an awesome post. :)
I didn't really understand abot carbon dating myself. I just believed it because... I believe in Science. ;)
Thanks I>T.
I wrote this to debunk Tim the Moron's claim that carbon dating doesn't work....in fact, carbon dating is very accurate because radiocarbon 14 decomposes at a very steady rate.
I like science (not that I'm any good at it,)I find thinking about scientific stuff calming. The emotional shit I'm involved in over at my blog is driving me nuts! But, you know what, dear beast? I'm not going to give up.
Fuck them trying to steamroller over me!
You are kinda mean to Tim, ya know.
I know.
But if you checked his comments at Atheist Revolution, he wasn't much kinder to the Atheists there (hence the ban by Vjack), and his obvious insistence to use Creationist material to justify his wacky beliefs, despite me refuting them again and again, is tiring.
Just think of me as a version of an anti-theist. I can't stand for religious morons who can't think.
Beast
Wow! I'm going to have to bookmark this one.
BTW, you've just been tagged with a blog meme: http://tinyurl.com/2td5l4
Enjoy!
Yeah, redundancy is annoying. the fault seems to be in part the mindset of religious people. They are not taught to think they are taught to not think. Consequently, they are ruled & fooled by their irrational emotions.
This whole way of NOT thinking is unfortunately, embedded in American culture.
& unfortunately, that is also why I can't link you back to my blog.
Very nice post, although it must be noted that Carbon Dating is not a fool proof method of dating whereby even Science guarantees all of its findings. Yes, I know your faithful saying, "potential falsifiability: Any scientific phenomenon or observation has the possibility of being proven false", and I understand Science to be an educated guess.
With that in mind there appears to be a few issues that need to be taken into consideration that could lead to the dates given by Science to be incorrect, noted not necessarily on purpose but may even be due to things out of their control.
1. Sample contamination
2. Variations in 14C content in the oceans
3. Fractionation effects
4. 14C concentration in the atmosphere
We must also consider that although adjustments can be made to try to include the problems that one may be face with, --“Without this historical environmental knowledge any attempt at carbon dating would be based on assumption (Stuiver, 1967; Morris, 1978; Morris, 1998). Therefore the accuracy of C-14 dating decreases with ages greater than 4000 BP [(before present)]. This decrease occurs because calibration ability is lost due to a lack of historical records (Brown, 1993)”-- but the assumptions themselves could be incorrect also causing further corrupt data.
Although one may disagree, if they are true to Science they must have an open mind that such errors are possible and have been found before in Science.
-----
As for your comments “But if you checked his comments at Atheist Revolution, he wasn't much kinder to the Atheists there (hence the ban by Vjack)”, I am not longer banned, and I shall let my posts here at your blog, and all of the atheists blogs I have visited, and how I have conversed with atheists at my blog to come in my defense.
Mr Splinter, Mr Splinter:
I think I have mentioned very clearly in my post:
1. The exponential rate of C-14 decay is very constant, with a tolerance of plus minus 40 years.
2. The discrepancies you have mentioned can be circumvented by using various calibration methods to create calibration charts to plot out very accurate ages of carbon dating.
Science doesn't stay stagnant, Mr Bullshiter. Only the bible does.
I actually do realize that you are no longer "banned" so to speak. All the better. I never supported it anyway. I prefer that you be allowed to pander your bullshit, so that I can refute them and irritate the shit out of you, and in the process inflate my own ego, so to speak.
Beast
hi, i have been looking up radioactive carbon dating recently to see how legit, and u found this article on ur "fav" site:
http://www.answersingenesis.org/articles/nab/does-c14-disprove-the-bible#fnList_1_7
At first, it seems like the Creationists have a good point, but it just doesn't smell right. I don't think scientists would just disregard something possible errors with carbon dating just to prove their points...that goes against the scientific dating. However, the above article still seems to raise questions, but I am not qualified to even look into it, haha. Could you please post something in reply to this article and its points in partiular, Beast? Thanks!
Hi Anon:
Usually I don't debunk stuff from AIG, because most of what is said, is, well.....bullshit.
Beast
So if you admit that Carbon Dating can only be truly effective up to 60,000 years and the "newest" dinosaurs died out over 1,000 times longer than that, even with calibration methods how do you expect to get a realistic figure with no point of reference to calibrate from. Their are over 600 active volcanoes on Earth and we have no idea what it was like then and with the release of carbon dioxide the plants take in less c-14 and the whole chain effect. That is like saying we can compare money from now to 100 years ago without any knowledge of inflation or gold standard.
There are other methods of radiometric dating:
* argon-argon (Ar-Ar)
* fission track dating
* helium (He-He)
* iodine-xenon (I-Xe)
* lanthanum-barium (La-Ba)
* lead-lead (Pb-Pb)
* lutetium-hafnium (Lu-Hf)
* neon-neon (Ne-Ne)
* optically stimulated luminescence dating
* potassium-argon (K-Ar)
* radiocarbon dating
* rhenium-osmium (Re-Os)
* rubidium-strontium (Rb-Sr)
* samarium-neodymium (Sm-Nd)
* uranium-lead (U-Pb)
* uranium-lead-helium (U-Pb-He)
* uranium-thorium (U-Th)
* uranium-uranium (U-U)
I understand there are dozens of ways to date, but again I go back to without a point of reference we are proposing we know the environment and atmospheric differences of millions of years. Scientific "facts" change almost daily as new things are discovered and these are present findings. Don't you find it kind of arrogant as a community to "guess" about things and propose them as fact.
Vince probably has not heard of "potential falsifiability".
Read this post before I explain further:
http://atheisthaven.blogspot.com/2007/06/scientific-jargon-why-christians-get-it.html
I have. I took Bio 181 just like everyone else in college and this quote "Any scientific phenomenon or observation has the possibility of being proven false," is exactly my point. The Theory of Evolution which I do know differs from evolution cannot be proven false nor true therefore making the whole argument a case of he said she said. No one was there and cannot "prove" anything. I am willing to let people teach The Theory of Evolution if they can open up their view points to realizing they don't know everything and there maybe another plausible yet again not provable beginning.
Vince:
Science is not about dogma; its about discovering the truth.
No one is saying evolution has to be 100% correct: The truth is we don't even know every form of life that has existed on this planet, let alone everything about the mechanics of life itself.
Beast
There is a bit of science that the writer of this article fails to grasp.
(Humor me for a moment) Nearly every culture has a story about a flood. Let's say there is something to this (or that at some time in earth's history the atmosphere was different) a canopy of water would have existed over the earth before the flood occurred. If that is the case less C-14 (the carbon dating measurement) would penetrate the field. Something prior to this canopy state would have a much smaller concentration of C-14. According to the current carbon dating method something dated prior to this event would appear much older than it really is.
It would be like someone trying to judge how long people were outside by their suntan. Just coming in was a man who was out in the blazing sun for 3 hours. He is burnt badly.
A second man was out earlier for 5 hours but there was heavy overcast the shielded the sun from him. He barely has a tan.
The man judging says the first man was out longer because his tan is brighter. The fact is the second was out longer, but because conditions were different his method of judging was wrong.
This story sums up a major flaw in the belief that carbon dating is accurate. Accept carbon dating works in reverse of the method used in the short story.
I think putting so much trust in this kind of science or technique is rather naive.
Carbon dating is quite frankly, a crock of shit. Here's the problems: You don't know when the Carbon became part of the structure you choose to date by measuring it. You don't know how or when the carbon molecule was created, nor do you know whether the rate of decay was constant (you assume it instead) because you can't measure the conditions in the past. You certainly can't take a time machine and measure it. You just have to 'assume' it which requires, *gasp*, blind faith. The only reason atheists, especially (I know, I used to be one), fight so hard for this ridiculous method is so you can try and hang on to the little threads of lies in order to prove the already 'disproven' theory of evolution to further the idea that 'God doesn't exist'. Ironically, it doesn't even take science to figure that out...only good sense.
As stated by others, the scientific communtiy assumes that the carbon ratio and the rate of decay in the past is the same as it is today.
It's like the dinosaurs: Scientists assume that they grew so large because they ate a lot, but do they ever consider that carbon and oxygen levels could have been MUCH higher back then?
Carbon dating is heavily flawed, and I see no proof that would lead me to believe that the ratio and amount of carbon present in the present day is the same as it was in the past.
Tim -
You're funny. You're going to sit here and dispute scientific methodology while at the same time expounding upon "a canopy of water would have existed over the earth"? Even if I assume you meant "a thick cloud layer", you do realize that atmosphere exists above the clouds right? Planes fly in it, using scientifically proven principles of engineering and aerodynamics. Yes, there are some clouds that exist at higher altitudes, but they don't produce condensation that reaches the earth.
That being said, cloud formations, do not significantly effect the type of radiation that forms 14C, since that formation occurs well inside the upper atmosphere.
Vince/Antero/Alex -
Seriously? We have environmental data going back millions of years that tells us exactly how much carbon was in the atmosphere. It comes from multiple different, yet corroborative sources, such as deep ice cores, deep ocean cores, speleothems, and dendrochronology. That's how we got the calibration curves in the first place.
Also, there is no scientific evidence of a "Great Flood". You have some misleading interpretations of data by creationists, along with a bunch of stories from around the world of *floods*, as if a flood was such a cosmically rare event that it needed supernatural explanation. I guess all the people in the mid-west can thank God, not that their lives were spared, but that their homes and livelihoods were all washed out in the the Mississippi.
So, if you're going to refute evidence accumulated by thousands of scientists in all kinds of different disciplines, try to come up with something better than stories.
One last thing. Don't try to refute a result by saying "we don't know what the conditions were 100,000 years ago". You already said that there was no "100,000 years ago". So, either it doesn't exist, or it existed and we don't know. Pick one and stick to it.
Interesting, I really liked this article and tend to agree with your findings. However, I did not agree with the generalizations made about Creationists as uneducated morons. I know some very smart Creationists I would be slow to get in an argument with.
I also was a little turned off by the language - not that I'm a prude, it just seemed unprofessional and definetely made me think twice about reading the article as an informative text. Sort of like something a "huckster" would have written.
you do realize the word circumvent means to avoid or bypass something by means of deception? Not very good choice of words to support a theory. Not only that but you do know the amount of carbon in the atmosphere has drastically changed in the past 11,000 years which affects a great deal of determining the age of a fossil.
May I point out a very important fact:
Creationism is not science.
Evolutionism is also not science.
They are merely a set of assumptions, based on one's own beliefs, on which data is interpreted.
For example, a piece of carbon dating data would actually be perfectly valid under both the Biblical-Creationist model as well as the Evolutionary model. But that data being interpreted by both models would give different dates. Note, same data, i.e. same proportions of C12 and C14 analysed, but different dates interpreted: Evolutionists would say some date, then the Creationists would say, "heck, I don't know."
K Flynn,
Let me rewrite your comment from a Creationist standpoint (please do not misunderstand me, no personal attack involved):
"You're funny. You're going to sit here and dispute scientific methodology while at the same time expounding upon "a primordial soup"? ... you do realize that atmosphere prohibits the formation of organic molecules right? Modern chemistry repeatedly tells us that, using scientifically proven principles... Yes, the Miller experiment in 1953 synthesised amino acids, but note that he used a cold trap to isolate those amino acids.
"... try to come up with something better than theories that are heavily revised every decade.
"One last thing. Don't try to support a result by saying "we do know what the conditions were 100,000 years ago". You already said that there a possiblity of error in the readings. So, either it has huge potential for error, or that we mere humans have God-like omniscience. Pick one and stick to it."
It should be clear by now that I am a Creationist myself. However, I do not view that Evolutionists are "moronic beasts." But it'd be nice if some of them could pause for a moment in the heat of the strife, and think a little bit more objectively (i.e. stop insulting for a while before continuing).
We Creationists should make sure we use accurate data before we launch an attack. There are many examples of both Creationists AND Evolutionists who talk crap because either of them have inaccurate facts on their hands.
For example, Tim, you don't accuse the writer for not being able to grasp science when you don't explain the "water canopy" model to him first. Of course everyone will disagree with you! Nobody (except people like me who already agrees with you) knows what the heck you're talking about.
Ahh so now I'm awaiting for someone to criticise me.
I know plenty of experiments that give inaccurate data because the experimenters took great care to eliminate errors caused by external factors: their mistake is that they miss out on some other factors they did not view as important. Anyone wants to fight with me in the field of musical instrument acoustics? I can give some very good examples in that field.
I hope this comment is objective enough. We should stop accusing everyone else of using deceitful language, being too emotionally (not spiritually, mind you, emotionally) propelled or just for being plain stupid.
Wow! I'm going to have to bookmark this one.
Beast,
You're an idiot.
Thanks!
Great discussion... I have to add that science needs to be blind as to the outcome... It seems that the author wants to believe something and tries to prove (or disprove the other side) with science. If the author wasn't so vitriolic towards his opposers and was truly impartial, it would give the argument more power. He sounds like the same "narrow-minded" zealots that he is against. I would bet that in the authors mind, there is no way that carbon dating is wrong and that bias (instantly disregarding a viable possibility) is not scientific at all. In fact the author is using faith to help his point while hiding behind science. Funny how he hates the other side for doing the same thing. At least the other side is open about their faith. Science does not need persuasion or hate, It stands on it's own. Unfortunatly for the author, carbon dating does not.
Although you have a good argument, the way you say it makes you look ignorant.
It is funny how L>T says he doesn't understand it, he just believes it. Isn't that what you claim that most Christians do? Just believing without understanding? It happens a lot in schools too where people just believe whatever they're taught without actually knowing how things work. We all have to accept the fact that there is so much that we don't know and that science will never explain. For example, the law of conservation of mass states that matter cannot be created or destroyed. If this is true, then where did everything come from? It had to be created by something, even if that wasn't a god. Just saying everyone should keep an open mind.
While it is incorrect to say that carbon dating doesn't work, it is equally incorrect to say that it does work. The reality is that carbon dating works with a high degree of accurate in very limited circumstances, such as when the original ratio of 14C to 12C is known OR when a highly reliable calibration is possible by having a secondary way to validate the age (e.g. dendrochronology). Radiocarbon dating can give us dates with a high degree of accuracy for land-dwelling plants and animals less than 4000 years old. Beyond that, the possible date ranges begin to diverge so wildly that other dating methods would be more appropriate.
to WKZH what the hell do you call biology then, ps beast you are AMAZING!!!
Part 1
Sorry to burst your bubble about carbon dating but its an inaccurate method of dating any artifact or remains. Every single theory of evolution is so far fetched and based on Darwin who himself never claimed it to be a fact but a theory which is really based on no real evidence, especially when we are now finding that carbon dating is not accurate. Time will always show mistakes, as technology advances. There is also the misconception that carbon dating can date millions of years when it can only date thousands. Also archaeologists do not regard 14C dates as absolute because of frequent anomalies, external factors like the environment can affect everything, thus speeding up the speed of decay. The amount of cosmic rays reaching the Earth will vary with the suns activity. As we can see today, there has been a huge increase in solar activity. Do we know what the environment was like back in ancient Egypt? Were the objects whatever they are, always in the same place or were they around a heat source or even a cold place? Sure, radiocarbon decomposes at a steady rate if the environment surrounding the object is around the same temperature. When someone is murdered, does the body always decay at the same rate if the environment is changed? If its in a hot place and then moved to a freezer or extremely cold place what would happen? Also its a well know fact that the Earths magnetic field has been decreasing, so more C than N is being produced today, than it has in the past. Maybe the conditions were more extreme and a lot more C was being produced. This shows that carbon dating is far off from being an accurate dating. People used to think that the flood is a fairytale, that 8 people did not get into an ark and that the earth was covered with water but as we now advance in time, the truth comes out more and more. We have hundreds of accounts of the flood, even the chinese have it with their words being symbols. The Bible has been criticized for years as being a fairytale with no historical value. Back in the days they had a field day disproving the Bible because there were no historical evidence to prove differently. As time goes on, more and more is being proven. For someone to think that absolutely nothing can create something, that life can come out of nothing is ludicrous. The perfection of our system is so complex, that one piece could collapse the entire system and life as we know it would not exist. Do you believe in alien life? Most scientists do, so the probability of us coming out of nothing is a number thats impossible, so imagine if there was other life forms, where did they come from? The same nothing as we did or their own nothing. lol. That number would increase with every life form that is introduced into the equation, to a number that would be incalculable.
Part 2
There is one last piece that I believe is important and that is the Genesis account of creation. We see that GOD created the earth in 6 days which can be argued that a day is like 1000 years with GOD. But even if it isnt. What was the time period between the 8th day until Adam and Eve were thrown out of the garden? The Bible teaches that we must have faith, so maybe that time period was left out on purpose. No one can prove that the Genesis account is wrong because even if the millions of years is correct in the dating, we can never say that the 8th day until the removal from the Garden is not millions or billions of years. Adam and Eve could not die in the garden, so any time outside of it is separate from when they were inside. The amount of years they lived outside does not become relevant in determining how long they lived, since death did not exist. Their years were counted outside the Garden. Did time exist in the same way before the fall of man or was it a day for 1000 years? All Im saying is that Science has in no way disproved the Genesis account because we can never know those days. The problem is that carbon dating is so inaccurate and the only reason people are atheist is because of these lies being used by the main stream scientific community. They used to say that Goliath was not a giant, that there were no such things as giants. lol. Again, as time goes on we see the truth comes out. I have never read in any magazine that they discovered the skeletal remains of humans who measured over 30 feet. For those who never heard of this, youre probably saying I'm crazy or its a lie. Stop allowing the mainstream media and science dictate to you, what is the facts or the truth. There is only one agenda for them and that is to control us and to discredit GOD ALMIGHTY. Anyone interested in science knows about patterns. Cant you see the pattern that the more time passes, the more we are finding out that the Bible is accurate and that the accounts of the Bible are not fairytales or lies. Many scientists today are starting to wake up and see that creation by a supreme being is possible and that the evidence is pointing to this fact. Faith is one of the biggest teachings in the Bible and what a better way to just accept on faith, that to not tell every little detail. When we look at everything, we see that its perfection is not the product of an accident out of nothingness, but in fact a very complex, and very intentional plan by a supreme being. I am not so sure that science is what GOD wanted from us. The Genesis account was about living on the earth in the garden and making it a beautiful place, expanding that very garden to cover the entire earth. This tells me that the rest of the earth might have been less habitable, that the environment was not a fertile as the garden. We were not supposed to eat from the tree of Knowledge. There are many facts and truths out there about the earth only being thousands of years and that carbon dating is in no way an accurate method. There is an accurate method which I will intentionally leave out,, so its up to you to search for it, unless you want to continue to live a lie or let others dictate whats the truth.
Part 3
Look up the giants and see how the mainstream media and science have hid this from you, how it was discovered years ago and if there was any reference made it was very minor. This is a huge discovery, literally. lol. Its not about being right or wrong people, its about the truth and lies that are being propagated to deceive and control us all, so that we can never reach our full potential, which GOD is the main part of our existence. Remember this. When someone says something that is truth, it will pierce you like a two edged sword, that is so sharp, it cuts to the bone. It releases anger, hate, cursing, putting people down like calling them moron etc. I have seen this so much, when someone heres the truth. I see it in a certain religion today, who would rather be right than live a truth or know that truth. Indoctrination is a very dangerous thing and I thank GOD I found that out, and learned everything with HIS help by believing and having faith. Everything in the Bible is coming true, and the time is very close. We just cannot continue on the path we are on. And science has not done us any real good when you really, honestly look and the whole picture. I will hope that you at least look into everything I have said. Its only a debate, its only a conversation. So when you get all pissed off and abusive because you hear something you dont like, ask yourself whats going on because I guarantee you, you heard truth. The truth hurts no matter what the topic is, love or science. Take care and GOD bless. If the idea of GOD was a lie, I would still rather believe in that and the product that comes out of that system than believe in science, which only destroys man and will never be used to truly help us. Just look at the world today. But, GOD does exist and the product of the system in the Bible, love, compassion, human family etc and trust in HIM through HIS Son Yehushua (Jesus), is a paradise on earth if we would just give up the lies and the grandios thinking. Take care.
I did forget one thing. The fact that the external factors like the environment would greatly increase any decay etc. The point I did not expand on was the part regarding the flood which there are around 500 flood stories in our history. Now we have someone like Charles Darwin who writes about evolution, which then takes people into this new belief system since they had no other argument that would disprove GOD, so they hang on to Darwin and build a net of lies around that theory. The flood alone, would severely change the environment. The earth was covered in water for centuries, not just 40 days and nights. The levels decreased in time which would have affected everything on this earth. The flood accounts are also a writing, like Darwin wrote so did they. But in the flood cases, there are 500 accounts of it happening. Darwin was one account which he explained at one time that its not a fact in any way. Even his wife said that he believed in a creator. Flooding can greatly alter sedimentation rates, so the original conditions cannot ever be known. How many other external factors have affected the environment? Could there have been other natural phenomenon or disasters in the past that we have no account of? The destruction of the Tower of Babel was something big, so who really know what happened right after the flooding which was still at high levels in those days. We can see that there is some truth to the destruction of the Babel. Theres the account of the Nephilim whos offspring became the giants, heros, etc. We never found these giants for many centuries so the story was dismissed. How about now where we see that there were giants in the days of the flood and tower of Babel. The sole purpose of the mainstream media and science is to prove anything that is against GOD or that gives up control to something other than governments, kings, leaders of any kind anywhere. Until man wakes up and sees the lies that are being told to us every single day by these outlets, we will never reach our true purpose together. If anyone says that the media or the government is there to protect us or tell us the truth, they are seriously out of touch with reality. Again, carbon dating is no where near being accurate, other than recent dating where all the information is available.
We just do not have all the information of the past and the one we do, the flood, can alter any environment which would alter any true dating. This is a fact that cannot be argued and if someone does argue this point they are seriously just lying to themselves or stubborn. If you know as much about carbon dating as you think you know that you must agree that the conditions and environment surrounding the object that is being tested is extremely important and that all the information regarding this must be included. If you want to think your god, than think it. If you just want to continue to think that you are in control than go ahead, but your bubble is about to burst because you arent as in control as you think. Just look at the governments of the world and even now the U.S is on the verge of being dismantled. The Constitution is being shredded as we speak. Why do these leaders hate the idea of GOD or creation? Maybe they are as evil as the Bible claims them to be, and the blind just wont ever see, they will continue to make excuses or ignore any facts. This will be received with anger and hate and cursing. Someone said on here that hes glad someone was back since he could push their buttons but reading their name calling I can see whos buttons are being pushed and whos getting angry at the truth. Its basic psychology and you can not ever push my buttons no matter how much your curse or call me names since my faith and love for GOD and all life is far greater than anything this world can produce. Especially lies or child like behavior. Man, you cant even have a debate any longer without someone trying to force their beliefs with threats of violence, anger, hate, name calling. This tells me a lot, well, everything. Take care and good luck on living with lies or being selective in what your research.
Of the scientists and engineers in the United States, only about 5% are creationists, according to a 1991 Gallup poll (Robinson 1995, Witham 1997). However, this number includes those working in fields not related to life origins (such as computer scientists, mechanical engineers, etc.). Taking into account only those working in the relevant fields of earth and life sciences, there are about 480,000 scientists, but only about 700 believe in "creation-science" or consider it a valid theory (Robinson 1995). This means that less than 0.15 percent of relevant scientists believe in creationism. And that is just in the United States, which has more creationists than any other industrialized country. In other countries, the number of relevant scientists who accept creationism drops to less than one tenth of 1 percent.
Additionally, many scientific organizations believe the evidence so strongly that they have issued public statements to that effect (NCSE n.d.). The National Academy of Sciences, one of the most prestigious science organizations, devotes a Web site to the topic (NAS 1999). A panel of seventy-two Nobel Laureates, seventeen state academies of science, and seven other scientific organizations created an amicus curiae brief which they submitted to the Supreme Court (Edwards v. Aguillard 1986). This report clarified what makes science different from religion and why creationism is not science.
One needs to examine not how many scientists and professors believe something, but what their conviction is based upon. Most of those who reject evolution do so because of personal religious conviction, not because of evidence. The evidence supports evolution. And the evidence, not personal authority, is what objective conclusions should be based on.
Often, claims that scientists reject evolution or support creationism are exaggerated or fraudulent. Many scientists doubt some aspects of evolution, especially recent hypotheses about it. All good scientists are skeptical about evolution (and everything else) and open to the possibility, however remote, that serious challenges to it may appear. Creationists frequently seize such expressions of healthy skepticism to imply that evolution is highly questionable. They fail to understand that the fact that evolution has withstood many years of such questioning really means it is about as certain as facts can get.
As you said, less than 0.15% of scientists believe in creation, and all of these panels and efforts are being made, and you're seriously trying to argue that evolution must be more credible because it has more evidence? At this point, it becomes a numbers game, literally tens of thousands more evolutionary scientists than creationists, and you're surprised that they are better supported? Now you're thinking "But scientists are objective, they must have looked for evidences to the contrary." Let me put it this way: how many of you evolutionists reading this have ever in your lives thought "I'm feeling objective today, I'm going to go look up evidence for creation?" I thought not. Now you're thinking, "It's not my job!" And now I ask, Whose is it? No evolutionary scientist will ever do this. (I say this without having studied every evolutionist to ever live.) Now, if you don't trust us under-educated creationists, consider this: you would rather trust people who spend their lives devoted to promoting their belief that life has no purpose and have been demonstrated to have covered up unfavourable evidence, over people who believe that lying is wrong and will earn you an eternal stay as the main course for the universe's largest barbecue.
Now I am just a layman, everyday person. I like carbon dating, it makes a lot of sense.
My question is: Is there anything that we know the age of which carbon dating has provided a correct age?
I have yet to read or find reliable results. If there are any I would love to see them.
But if the theory does not hold up to testing, it is not a strong theory which is good science.
For the truth about carbon dating from real scientist that dont have an agenda to move God from this world AT ALL COSTS, go to www.focuspress.org or google scientist Brad Harrub. And then some other guy above says there is no evidence for the flood...lol where do u think all the fossils came from...and do u really believe the grand canyon was cut by the Colorado River,...lol..I guess Iif you will believe the hoax of evolution youll believe anything. God and real science are a perfict fit. Please just listen to both sides...they are all scientists there are thousands of scientist who know evolution is a joke, its just a huge conspiracy and a bunch of ego maniacs that are scared to admit their wrong...they will go through great length to falsify their fake evidences. So much evidence points to a young earth...just examine the evidence for yiurself and dont take these liars word for it.. what about polly straight fossils... and why is there sea fossils at the top of every mountain, how did they get there? Evolutionst dont like mount saint helens...focuspress.org tells you why....check out Lee Stroble an honest atheist who examined the evidence himself because what his buddys where saying just didnt add up...they would tell them just go with it...check out Ben Stiens "expelled" documentary and see the big conspiracy...youll get fired for just examining creation...or mentioning it...this evolution crap is probably the biggest hoax of allmankind....even darwin said it had alot of issues but someday they would have the missing link... I guess a dot the size of period that came from nothing, blew up in outer space and turned into everything you see today...kangaroos, giraffs, skunks, strawberrys, cherrys, apples, oranges, graapes, watermelon...red...yellow, green....octopus...whales...ants...bees...LOL... did u know if the sun was just fractions in distance from earth closer we would burn up...further we would freeze... did u know its the same formthe moon...if the moon were further away by fractions there would be no movement in the ocean and we wouldn't have enough oxygen. Also the moon is moving further from the earth...even if it started right next to the earth in just millions of years it would be so far away life couldnt exsist...there is just so so so much facts like this that destroys the old earth idea and evolution... face it evolution is just another false religion like islam, Catholicism...mormans...and so on... remember the bible even says that this very thing would happen 2 Timothy 4:3...and dont let anyone tell u there are any...yes any contridictions in the bible..they r so called contridictions...just taking things out of context or misunderstanding translations...example...one verse says Judas hung himself but another says he fell forward and his guts burst open....well he did hang himself as one writer mentioned...the next writer says talks about him falling forward and bursting open...well judas hung himself, decomposed got bloated and the rope broke he landed on his feet and fell foward and burst open...both happened...simple explanation but blind foolish atheist will say see the bible is full of contradictions... the fact is it was written by many different people over a span of 1500 years from people of different race and regions yet it falls perfectly in place and all prophecy has came true except the ones yet to happen... hit me up if u wanna know more. Seektruth41@hotmail.com in Christ!
'Nuff said. Make your choice. I choose to believe that there is a God and he created the world around us. Dispute that.
Just a suggestion, but nearly everyone who took part in this discussion (and most especially the author of the "article") might want to consider enrolling in a Freshman-level course in Critical Thinking.
The ignorance on display here is quite astounding. For such supposedly intelligent individuals to demonstrate a complete lack of ability to construct a sound and coherent argument supported by citable research is laughable. You folks "know" far less than you think you do.
Science and the world in general would be so much better off if the education of the masses was left soley to capable individuals with a distinct lack of any personal agendas.
Truth only sets free those who don't run away from it because they don't like it.
This is full of holes as the one you tried to debunk. Probably more.
Anything that has error, even just a small amount cannot be called hard proof for science. Even if it is just a possibility of error. Sorry. That's just it. It's never accurate if there is a single anomaly in the process.
Carbon dating is never accurate. PERIOD.
Being the intellectual equivalent of hucksters and mobsters, scientists of these sort are apt to criticize the Holy Scriptures in a bid to construe evidence to fit into their pseudo-scientific bullshit. They sift through biblical literature, pick the parts that supposedly criticize a creationist belief, and put up claims that mainstream theosophy is bogus.
These deranged fundamentalists are not interested in biblical truths; they don't carry out any field work or research. All they ever do is throw around their banter and hope that someone, somewhere, will lap up and believe in their Darwinist beliefs, which coincidentally, have no scientific merit whatsoever.
Great post! Been reading about carbon testing a lot recently. Great info, thanks!
So my choices are to follow the evolutionist and refute anything supernatural(to what end?) or follow the teachings of God's word and live in love and harmony with my brothers and sisters and nature until we make it to paradise. Not a tough choice for me :)
Let love in brothers and sisters.
Radiocarbon dating can easily establish that humans have been on the earth for over twenty thousand years, at least twice as long as creationists are willing to allow. Therefore it should come as no surprise that creationists have been trying desperately to discredit this method for years. They have their work cut out for them, however, because radiocarbon (C-14) dating is one of the most reliable of all the radiometric dating methods.
I will answer several of the most common creationist attacks on carbon-14 dating.
Question: How does carbon-14 dating work?
Answer: Cosmic rays in the upper atmosphere are constantly converting the isotope nitrogen-14 (N-14) into carbon-14 (C-14 or radiocarbon). Living organisms are constantly incorporating this C-14 into their bodies along with other carbon isotopes. When the organisms die, they stop incorporating new C-14, and the old C-14 starts to decay back into N-14 by emitting beta particles. The older an organism's remains are, the less beta radiation it emits because its C-14 is steadily dwindling at a predictable rate. So, if we measure the rate of beta decay in an organic sample, we can calculate how old the sample is. C-14 decays with a half-life of 5,730 years.
Question: Kieth and Anderson radiocarbon-dated the shell of a living freshwater mussel and obtained an age of over two thousand years. ICR creationists claim that this discredits C-14 dating. How do you reply?
Answer: It does discredit the C-14 dating of freshwater mussels, but that's about all. Kieth and Anderson show considerable evidence that the mussels acquired much of their carbon from the limestone of the waters they lived in and from some very old humus as well. Carbon from these sources is very low in C-14 because these sources are so old and have not been mixed with fresh carbon from the air. Thus, a freshly killed mussel has far less C-14 than a freshly killed something else, which is why the C-14 dating method makes freshwater mussels seem older than they really are. When dating wood there is no such problem because wood gets its carbon straight from the air, complete with a full dose of C-14. The creationists who quote Kieth and Anderson never tell you this, however.
Question: A sample that is more than fifty thousand years old shouldn't have any measurable C-14. Coal, oil, and natural gas are supposed to be millions of years old; yet creationists say that some of them contain measurable amounts of C-14, enough to give them C-14 ages in the tens of thousands of years. How do you explain this?
Answer: Very simply. Radiocarbon dating doesn't work well on objects much older than twenty thousand years, because such objects have so little C-14 left that their beta radiation is swamped out by the background radiation of cosmic rays and potassium-40 (K-40) decay. Younger objects can easily be dated, because they still emit plenty of beta radiation, enough to be measured after the background radiation has been subtracted out of the total beta radiation. However, in either case, the background beta radiation has to be compensated for, and, in the older objects, the amount of C-14 they have left is less than the margin of error in measuring background radiation. As Hurley points out:
Without rather special developmental work, it is not generally practicable to measure ages in excess of about twenty thousand years, because the radioactivity of the carbon becomes so slight that it is difficult to get an accurate measurement above background radiation. (p. 108)
Cosmic rays form beta radiation all the time; this is the radiation that turns N-14 to C-14 in the first place. K-40 decay also forms plenty of beta radiation. Stearns, Carroll, and Clark point out that ". . . this isotope [K-40] accounts for a large part of the normal background radiation that can be detected on the earth's surface" (p. 84). This radiation cannot be totally eliminated from the laboratory, so one could probably get a "radiocarbon" date of fifty thousand years from a pure carbon-free piece of tin. However, you now know why this fact doesn't at all invalidate radiocarbon dates of objects younger than twenty thousand years and is certainly no evidence for the notion that coals and oils might be no older than fifty thousand years.
Question: Creationists such as Cook (1966) claim that cosmic radiation is now forming C-14 in the atmosphere about one and one-third times faster than it is decaying. If we extrapolate backwards in time with the proper equations, we find that the earlier the historical period, the less C-14 the atmosphere had. If we extrapolate as far back as ten thousand years ago, we find the atmosphere would not have had any C-14 in it at all. If they are right, this means all C-14 ages greater than two or three thousand years need to be lowered drastically and that the earth can be no older than ten thousand years. How do you reply?
Answer: Yes, Cook is right that C-14 is forming today faster than it's decaying. However, the amount of C-14 has not been rising steadily as Cook maintains; instead, it has fluctuated up and down over the past ten thousand years. How do we know this? From radiocarbon dates taken from bristlecone pines.
There are two ways of dating wood from bristlecone pines: one can count rings or one can radiocarbon-date the wood. Since the tree ring counts have reliably dated some specimens of wood all the way back to 6200 BC, one can check out the C-14 dates against the tree-ring-count dates. Admittedly, this old wood comes from trees that have been dead for hundreds of years, but you don't have to have an 8,200-year-old bristlecone pine tree alive today to validly determine that sort of date. It is easy to correlate the inner rings of a younger living tree with the outer rings of an older dead tree. The correlation is possible because, in the Southwest region of the United States, the widths of tree rings vary from year to year with the rainfall, and trees all over the Southwest have the same pattern of variations.
When experts compare the tree-ring dates with the C-14 dates, they find that radiocarbon ages before 1000 BC are really too young—not too old as Cook maintains. For example, pieces of wood that date at about 6200 BC by tree-ring counts date at only 5400 BC by regular C-14 dating and 3900 BC by Cook's creationist revision of C-14 dating (as we see in the article, "Dating, Relative and Absolute," in the Encyclopaedia Britannica). So, despite creationist claims, C-14 before three thousand years ago was decaying faster than it was being formed and C-14 dating errs on the side of making objects from before 1000 BC look too young, not too old.
Question: But don't trees sometimes produce more than one growth ring per year? Wouldn't that spoil the tree-ring count?
Answer: If anything, the tree-ring sequence suffers far more from missing rings than from double rings. This means that the tree-ring dates would be slightly too young, not too old.
Of course, some species of tree tend to produce two or more growth rings per year. But other species produce scarcely any extra rings. Most of the tree-ring sequence is based on the bristlecone pine. This tree rarely produces even a trace of an extra ring; on the contrary, a typical bristlecone pine has up to 5 percent of its rings missing. Concerning the sequence of rings derived from the bristlecone pine, Ferguson says: In certain species of conifers, especially those at lower elevations or in southern latitudes, one season's growth increment may be composed of two or more flushes of growth, each of which may strongly resemble an annual ring. Such multiple growth rings are extremely rare in bristlecone pines, however, and they are especially infrequent at the elevation and latitude (37� 20' N) of the sites being studied. In the growth-ring analyses of approximately one thousand trees in the White Mountains, we have, in fact, found no more than three or four occurrences of even incipient multiple growth layers. (p. 840)
In years of severe drought, a bristlecone pine may fail to grow a complete ring all the way around its perimeter; we may find the ring if we bore into the tree from one angle, but not from another. Hence at least some of the missing rings can be found. Even so, the missing rings are a far more serious problem than any double rings.
Other species of trees corroborate the work that Ferguson did with bristlecone pines. Before his work, the tree-ring sequence of the sequoias had been worked out back to 1250 BC. The archaeological ring sequence had been worked out back to 59 BC. The limber pine sequence had been worked out back to 25 BC. The radiocarbon dates and tree-ring dates of these other trees agree with those Ferguson got from the bristlecone pine. But even if he had had no other trees with which to work except the bristlecone pines, that evidence alone would have allowed him to determine the tree-ring chronology back to 6200 BC. (See Renfrew for more details.)
So, creationists who complain about double rings in their attempts to disprove C-14 dating are actually grasping at straws. If the Flood of Noah occurred around 3000 BC, as some creationists claim, then all the bristlecone pines would have to be less than five thousand years old. This would mean that eighty-two hundred years worth of tree rings had to form in five thousand years, which would mean that one-third of all the bristlecone pine rings would have to be extra rings. Creationists are forced into accepting such outlandish conclusions as these in order to jam the facts of nature into the time frame upon which their "scientific" creation model is based.
Question: Creationist Thomas G. Barnes has claimed that the earth's magnetic field is decaying exponentially with a half-life of fourteen hundred years. Not only does he consider this proof that the earth can be no older than ten thousand years but he also points out that a greater magnetic strength in the past would reduce C-14 dates. Now if the magnetic field several thousand years ago was indeed many times stronger than it is today, there would have been less cosmic radiation entering the atmosphere back then and less C-14 would have been produced. Therefore, any C-14 dates taken from objects of that time period would be too high. How do you answer him?
Answer: Like Cook, Barnes looks at only part of the evidence. What he ignores is the great body of archaeological and geological data showing that the strength of the magnetic field has been fluctuating up and down for thousands of years and that it has reversed polarity many times in the geological past. So, when Barnes extrapolates ten thousand years into the past, he concludes that the magnetic field was nineteen times stronger in 4000 BC than it is today, when, actually, it was only half as intense then as now. This means that radiocarbon ages of objects from that time period will be too young, just as we saw from the bristlecone pine evidence.
Question: But how does one know that the magnetic field has fluctuated and reversed polarity? Aren't these just excuses scientists give in order to neutralize Barnes's claims?
Answer: The evidence for fluctuations and reversals of the magnetic field is quite solid. V. Bucha, a Czech geophysicist, has used archaeological artifacts made of baked clay to determine the strength of the earth's magnetic field when they were manufactured. He found that the earth's magnetic field was 1.5 times as strong as today around 1 AD, 1.6 times as strong around 400 BC, 0.8 times as strong around 2000 BC, and only 0.5 times as strong around 4000 BC. (See Bailey, Renfrew, and Encyclopedia Britannica for details.) In other words, it rose in intensity from 0.5 times its present value in 4000 BC to a peak of 1.6 times its present value in 400 BC, and it has been slowly declining since then. Even before the bristlecone pine calibration of C-14 dating was worked out by Ferguson, Bucha predicted that this change in the magnetic field would make radiocarbon dates too young.
This idea [that the fluctuating magnetic field affects influx of cosmic rays, which in turn affects C-14 formation rates] has been taken up by the Czech geophysicist, V. Bucha, who has been able to determine, using samples of baked clay from archeological sites, what the intensity of the earth's magnetic field was at the time in question. Even before the tree-ring calibration data were available to them, he and the archeologist, Evzen Neustupny, were able to suggest how much this would affect the radiocarbon dates. (Renfrew, p. 76)
Not only that, but his predictions were confirmed in detail:
There is a good correlation between the strength of the earth's magnetic field (as determined by Bucha) and the deviation of the atmospheric radiocarbon concentration from its normal value (as indicated by the tree-ring radiocarbon work). (Renfrew, p. 76)
So, once we know all the magnetic data, we see that it really supports the tree-ring calibration of C-14 dating, rather than the conclusions of Cook and Barnes.
As for the question of polarity reversals, plate tectonics can teach us much. It is a fact that new oceanic crust continually forms at the mid-oceanic ridges and spreads away from those ridges in opposite directions. When lava at the ridges hardens, it keeps a trace of the magnetism of the earth's magnetic field. Therefore, every time the magnetic field reverses itself, bands of paleomagnetism of reversed polarity show up on the ocean floor alternated with bands of normal polarity. These bands are thousands of kilometers long, they vary in width, they lie parallel, and the bands on either side of any given ridge form mirror images of each other. Thus it can be demonstrated that the magnetic field of the earth has reversed itself dozens of times throughout earth history.
Barnes, writing in 1973, ought to have known better than to quote the gropings and guesses of authors of the early sixties in an effort to debunk magnetic reversals. Before plate tectonics and continental drift became established in the mid-sixties, the known evidence for magnetic reversals was rather scanty, and geophysicists often tried to invent ingenious mechanisms with which to account for this evidence rather than believe in magnetic reversals. However, by 1973, sea floor spreading and magnetic reversals had been documented to the satisfaction of almost the entire scientific community. Yet, instead of seriously attempting to rebut them with up-to-date evidence, Barnes merely quoted the old guesses of authors who wrote before the facts were known. But, in spite of Barnes, paleomagnetism on the sea floor conclusively proves that the magnetic field of the earth oscillates in waves and even reverses itself on occasion. It has not been decaying exponentially as Barnes maintains.
Question: Does outside archaeological evidence confirm theC-14 dating method?
Answer: Yes. When we know the age of a sample through archaeology or historical sources, the C-14 method (as corrected by bristlecone pines) agrees with the age within the known margin of error. For instance, Egyptian artifacts can be dated both historically and by radiocarbon, and the results agree. At first, archaeologists used to complain that the C-14 method must be wrong, because it conflicted with well-established archaeological dates; but, as Renfrew has detailed, the archaeological dates were often based on false assumptions. One such assumption was that the megalith builders of western Europe learned the idea of megaliths from the Near-Eastern civilizations. As a result, archaeologists believed that the Western megalith-building cultures had to be younger than the Near Eastern civilizations. Many archaeologists were skeptical when Ferguson's calibration with bristlecone pines was first published, because, according to his method, radiocarbon dates of the Western megaliths showed them to be much older than their Near-Eastern counterparts. However, as Renfrew demonstrated, the similarities between these Eastern and Western cultures are so superficial that the megalith builders of western Europe invented the idea of megaliths independently of the Near East. So, in the end, external evidence reconciles with and often confirms even controversial C-14 dates.
One of the most striking examples of different dating methods confirming each other is Stonehenge. C-14 dates show that Stonehenge was gradually built over the period from 1900 BC to 1500 BC, long before the Druids, who claimed Stonehenge as their creation, came to England. Astronomer Gerald S. Hawkins calculated with a computer what the heavens were like back in the second millennium BC, accounting for the precession of the equinoxes, and found that Stonehenge had many significant alignments with various extreme positions of the sun and moon (for example, the hellstone marked the point where the sun rose on the first day of summer). Stonehenge fits the heavens as they were almost four thousand years ago, not as they are today, thereby cross-verifying the C-14 dates.
Question: What specifically does C-14 dating show that creates problems for the creation model?
Answer: C-14 dates show that the last glaciation started to subside around twenty thousand years ago. But the young-earth creationists at ICR and elsewhere insist that, if an ice age occurred, it must have come and gone far less than ten thousand years ago, sometime after Noah's flood. Therefore, the only way creationists can hang on to their chronology is to poke all the holes they can into radiocarbon dating. However, as we have seen, it has survived their most ardent attacks.
Bailey, Lloyd R. 1978. Where Is Noah's Ark? Nashville, TN: Abington Press.
Barnes, Thomas G. 1973. Origin and Destiny of the Earth's Magnetic Field. San Diego: Creation-Life Publishers.
Cook, Melvin A. 1966. Prehistory and Earth Models. London: Max Parrish and Co., Ltd.
"Dating, Relative and Absolute." Encyclopaedia Britannica: Macropaedia, Vol. 5. 1974.
"Earth, Magnetic Field of." Encyclopaedia Britannica: Macropaedia, Vol. 5. 1974.
Fergusson, C. W. 1968. "Bristlecone Pine: Science and Aesthetics." Science 159:839-846.
Hawkins, Gerald S. 1965. Stonehenge Decoded. New York: Doubleday & Co.
Hurley, Patrick M. 1959. How Old Is the Earth? New York: Doubleday & Co.
Kieth, M. C., and Anderson, G. M. August 16, 1963. "Radiocarbon Dating: Fictitious
Results with Mollusk Shells." Science 141:634ff.
Kofahl, Robert E. 1977. The Handy Dandy Evolution Refuter. San Diego: Beta Books.
Morris, Henry M. (ed.) 1974. Scientific Creationism. San Diego: Creation-Life Publishers.
Renfrew, Colin. 1973. Before Civilization. New York: Alfred Knopf.
Slusher, Harold S. 1973. Critique of Radiometric Dating. San Diego: Creation-Life Publishers.
Stearns, Colin W., Carroll, Robert L., and Clark, Thomas H. 1979. Geological Evolution of North America, 3rd Edition. New York: John Wiley &
If it the stuff on the AIG websites is such nonsense and you claim to be so intelligent why don't you try "debunking it" the fact of the matter is evolution outside of a species or macro-evolution is a process that has never been scientifically reproduced through experiment (i.e. fruit fies, even bacteria,) there may have been diversity within the limitations of those species found but never has a diversity been found that rexalted in new genetic information thus creating a completely new species further the mutations that have been found have only been harmfull mutations particularly in fruit flies so that when the breeds with the mutations are realised "into the wild" they very quickly die off so maybe the reason people have an issue with evolution isn't so much that they are stupid or uniformed, maybe perhaps it is that macro-evolution has never been reproduced through experiment which is the case for most "science" which is widely accepted as fact, maybe it's because evolution is not science at all maybe it's pretty much guess work like anything else and noone can come up with the answers to where life originated instead they just confuse people and hope they never ask the real question which the evolution cannot answer "where did the first forms if life come from?
No they figure if theyou guess enough times and present it as fact people will stop arguing lol that's the atheist logic
https://m.youtube.com/watch?v=KK3eh4Z5Ko4
Educate yourselves. Christians do not hate or ignore sound science. We use science to prove the Bible true. We love science. God created science.
Cotradiction -" matter can't be *created or destroyed. It had to be *created by something." If matter can't be created why say it was created?
Read the statement you replied to a bit more thoroughly. Joel was saying "the law of conservation of mass states that matter cannot be created or destroyed" and that "law" doesn't make much sense when you consider theories such as the big bang theory, all matter crammed down into the size of an atom or a neutron or smaller still. Perhaps I got carried away, just read, think, re-read, then understand, then post, there's my $0.02.
Post a Comment